A “healthy brain” is a brain in its neural dynamic stable state. Deviation from neural cohesion and symmetry is usually a result of the brain’s exposure to trauma, either physical traumatic brain injury or psychogenic. Such trauma can trigger sleep disturbance, insomnia, post-traumatic stress disorder, anxiety, depression, bipolar disorder, and other mental disorders, which are often subsequently self-medicated by those affected, compounding the changes in the brain’s physiological condition and negatively impacting an individual’s behavior.
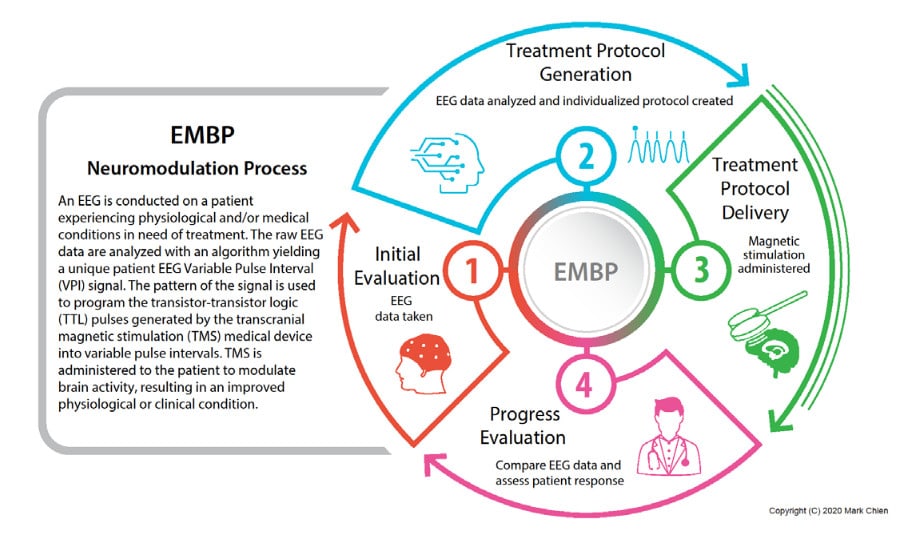
In addition to behavioral evaluations, many scientific tests are available to directly measure the brain’s physiological properties regarding its health. Studies with co-registered assessments of Positron Emission Tomography (PET) scan, Magnetic Resonance Imaging (MRI), and Electroencephalogram (EEG) have shown that the brain metabolic rate is determined, to a great extent, by the levels of symmetry in its underlying electrical activities. Electromagnetic Brain Pulsing (EMBP) is a new, advanced, and patented technology.